Is It Beneficial to Eat Soil?

The rates of infectious diseases that are unresponsive to the antibiotics used to treat them continue to skyrocket. Drug resistance and super bugs abound. Simply stated, drug resistance results when infection-causing bacteria, fungi, parasites or viruses evolve and become immune to the drugs once used to destroy them.
Data from the U.S. Centers for Disease Control and Prevention (CDC)1 suggests about 2 million Americans are diagnosed annually with infections known to be unresponsive to antibiotics, resulting in the deaths of 23,000 people. In Europe, 25,000 people die each year from drug-resistant infections.2 Worldwide, at least 700,000 die each year from drug resistance associated with illnesses such as bacterial infections, HIV, AIDS, malaria and tuberculosis.3
Now, a study published in Nature Microbiology4 highlights a hopeful discovery in the battle to fight back against super bugs: After studying 2,000 soil samples, researchers from two prominent U.S. universities have uncovered a potential new class of antibiotics. Surprisingly it was found in dirt. Their discovery not only highlights the potential for one or more new bacteria-killing drugs, but also begs the question, is it beneficial to eat soil?
Scientists Discover Potential New Class of Antibiotics in Dirt
In the process of discovering a potential new class of antibiotics, researchers from New York’s Rockefeller University and New Jersey-based Rutgers University were able to extract, clone and sequence DNA from soil samples.5 Their objective was to identify any genes able to produce molecules with antibiotic potential. After systematically searching through hundreds of soil samples, they discovered a new antibiotic class called malacidins.
It seems, the researchers say, malacidins may be able to attack and kill many types of super bugs, including the dreaded methicillin-resistant Staphylococcus aureus (MRSA). In tests involving rats infected with MRSA, malacidins were able to successfully sterilize cuts in their skin.
Remarkably, after three weeks of exposure, the bacterium showed zero signs of resistance.6 While this is not the first time antibiotics have been extracted from soil, it is the first time scientists have successfully identified a bacterial species with the potential to become a drug.
As with the discovery of penicillin, the world’s first antibiotic, most bacteria-fighting drugs have been revealed through lab-based tests. Now, instead of growing antibiotics in petri dishes, scientists like Sean Brady, Ph.D., associate professor at Rockefeller University and head of their Genetically Encoded Small Molecules lab, are moving their research outdoors.
“Every place you step, there’s 10,000 bacteria, most of which we’ve never seen,” said Brady. “Our idea is, there’s this reservoir of antibiotics out in the environment we haven’t accessed yet.”7
While it takes years for molecules like malacidins to be developed, tested and approved for use as a medication, the discovery of these powerful soil microbes is a hopeful sign. The introduction of malacidins highlights the value of pursuing natural solutions as a means of helping counter the growing epidemic of drug-resistant infections.
Brady and his fellow researchers have cause for celebration, particularly given the fact science hasn’t brought forth “a truly new antimicrobial medication since 1987.”8 The discovery of malacidins, he says, “is proof of a powerful principle: A world of potentially useful untapped biodiversity is still waiting to be discovered.”9
The Keys to Bringing Forth Malacidin, ‘Killer of Bad Guys’
According to the Los Angeles Times,10 malacidins, as a potential new class of antibiotics, will do well if they are able to live up to the rough Latin translation of their name: “killer of bad guys.” Malacidins, they suggest, is short for metagenomic acidic lipopeptide antibiotic-cidins. Additionally, “mal” means bad in Latin, and “cide” means to kill. Notably, malacidins are a distant relative of the powerful antibiotic daptomycin, which was introduced in 2003 and, to date, remains unchallenged by resistant bacteria.
Daptomycin is known to use calcium to disrupt bacterial cell walls. While malacidins appear to work differently than daptomycin, scientists believe they will be at least as effective given they were unable to be thwarted by resistant bacteria after 20 days of continuing contact. Typically, this period of time would be sufficient for most bacteria to find a way to overcome an antibiotic’s effects.
The fact the majority of microbes with antibiotic properties don’t grow well under controlled laboratory conditions makes them difficult to study. To speed up their efforts, Brady and his team leveraged metagenomics, a term coined in the late ‘90s to describe the practice of sequencing genetic material recovered directly from environmental samples en masse.
It involves cloning vast quantities of DNA from hundreds of soil samples and sorting through the material in an orderly manner in hopes of uncovering interesting sequences. About the work, the Los Angeles Times stated:11
“Brady’s team was looking specifically for distant relatives of daptomycin, which uses calcium to bust up, break down and generally disrupt the cell walls of target bacteria. They knew that long after the effectiveness of other antibiotics has waned, daptomycin continued to kill its targets, and they surmised that its distinctive use of calcium might be the key to an antibiotic compound’s longevity.
They also knew that trying to culture all their soil samples in a lab would take forever, and that most would not replicate themselves under lab conditions anyway. So instead, they used high-speed computer processing to ‘screen’ the soil samples for the distinctive chemical hallmark of calcium dependence.
When they found what they were looking for in a particular sample of desert soil, they captured and cloned the relevant genes, rearranged and inserted them into a host organism, and expanded the resulting sample through fermentation. This process made it possible to test the unique properties of malacidins on MRSA-infected rats.”
Brady and company are not the only ones hoping to accelerate the discovery process for potential new medications able to treat disease. Metagenomics has also been used to seek out new antibiotics in environments ranging from insect guts to ocean water.12 Furthermore, the technique has revealed the extent of antibiotic resistance based on studies of polluted lakes and urban sewage.13,14
What Causes Antibiotic Resistance?
The rush to identify new antibiotic drugs continues to be fueled by rising levels of drug resistance. The trouble starts when antibiotics kill not only the bad bacteria causing your illness but also your good gut bacteria — the ones needed to help protect your body from infection. With less good bacteria to fight back, the drug-resistant bacteria are able to grow stronger and take over. Even worse, some of the bad bacteria pass along their drug resistance abilities to other bacteria, exacerbating the problem.
This is, in part, why it is important to replenish your gut bacteria after completing a course of antibiotics. You need to rebalance your intestinal flora. Using even a single round of antibiotics can contribute to the development of drug resistance, especially because many prescribed antibiotics are unnecessary and cause damage. For this reason, you should use antibiotic drugs only when necessary to treat bacterial infections.
That said, do not bother taking antibiotics to fight the common cold or flu because your body will recover from viral infections only after the illness has run its course. Taking antibiotics for viral infections is both unnecessary and dangerous since it contributes to the problem of drug resistance. About antibiotics, the CDC states:15
“Antibiotics are among the most commonly prescribed drugs used in human medicine, and can be lifesaving drugs. However, up to 50 percent of the time, antibiotics are not optimally prescribed, often done so when not needed [and given with] incorrect dosing or duration.”
As shown in the diagram below, other major factors in the growth of antibiotic resistance are the spread of resistant strains of bacteria from person to person or from animals to people through the food supply. Beyond the human abuse of antibiotics, the pervasive misuse of these drugs by the agriculture industry plays a significant role.
After all, nearly 80 percent of all antibiotics used in the U.S. are applied within concentrated animal feeding operations (CAFOs), making them a clearinghouse for antibiotic-resistant disease. These factory farms breed antibiotic-resistant bacteria due to their long-standing practice of continuously administering low doses of antibiotics to CAFO animals, which enables pathogens to survive, adapt and eventually thrive.
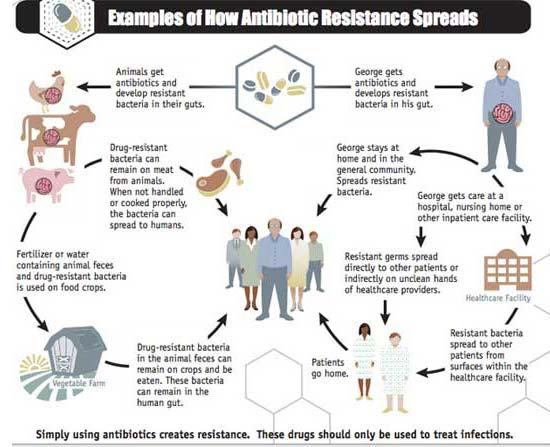
Source: CDC.gov, Examples of How Antibiotic Resistance Spreads, 2013
Tips on Fighting Antibiotic Resistance
You can help fight antibiotic resistance by choosing to:
- Avoid eating meat, eggs or dairy products sourced from CAFOs; find a local source for organic grass fed meat and dairy
- Consume probiotics and/or sporebiotics during and immediately following a course of antibiotics to replenish your healthy gut flora
- Decline antibiotics offered to treat viral infections even when prescribed by your doctor — they will have zero effect
- Eat fermented foods, such as kimchi, natto and yogurt, as well as prebiotic foods from fiber-rich sources such as asparagus, garlic and onions to promote healthy gut bacteria
- Reserve antibiotic use for the treatment of bacterial infections only
Sporebiotics Stand up to Antibiotics, Aid in Resolving Food Intolerances
Sporebiotics, or spore-based probiotics, are an excellent complement to regular probiotics. They are part of a group of derivatives of the microbe called bacillus, a genus with hundreds of subspecies, the most important of which is bacillus subtilis. Sporebiotics consist of the cell wall of bacillus spores, and they are a primary tool to boost your immune tolerance. Because sporebiotics do not contain any live bacillus strains — only its spores — they are unaffected by antibiotics.
Because antibiotics indiscriminately kill your gut bacteria, both the good and bad kinds, secondary infections and lowered immune function are among the common side effects you may experience when taking such drugs. Fortunately, sporebiotics can help fortify your gut microbiome since they can withstand antibiotics.
In addition, sporebiotics are resilient and able to survive passage through your stomach acid. In contrast, many acidophilus products are killed off by your stomach acid, rendering them ineffective. Once established in your gut, sporebiotics help improve your intestinal barrier function. This bodily function determines which nutrients to absorb and which to excrete.
Spores also increase your immune tolerance, which means they help repair damage in your intestinal barrier from conditions such as leaky gut. My longtime mentor Dr. Dietrich Klinghardt, who also holds a Ph.D. and is founder of the Klinghardt Academy in Washington (state), advocates the use of sporebiotics for the treatment of food intolerances related to ALS (amyotrophic lateral sclerosis), autism, Lyme disease, multiple sclerosis and Parkinson’s disease.
Could You Benefit From More Contact With (Healthy) Dirt?
If you live in a developed country, you may be among those who make the mistake of associating good health with cleanliness. Getting your hands dirty, particularly while outdoors, may happen only on rare occasions and generally is frowned upon. In an effort to “fight germs,” so-called modern homes — as well as the people living in them — are scrubbed clean with antibacterial soaps and other antibacterial household products. Sadly, this overfocus on cleanliness disconnects you from the very soil that is vital to your survival.
Rather than safeguard health, chemical-infused hygiene tactics serve to wipe out many of the microorganisms to which your body needs exposure to develop and maintain proper immune function. Trying to instill good hygiene for yourself and your family members through a lack of exposure to dirt, germs, parasites and even viruses may actually have a noticeably opposite and somewhat negative effect.
Rather than develop a healthy intolerance to “germy” environmental influences, you may actually become more vulnerable to illnesses and disease. After all, at least one expert suggests allowing small children to pick their noses and eat their boogers may actually be a good thing! In terms of indoor cleanliness (even after nose picking), I recommend soap and water for hand-washing.
Avoid the antibacterial chemicals (typically triclosan) found in hand sanitizers and hand soap. Antibacterial agents are quite toxic and have even been found to promote the growth of resistant bacteria.
With respect to outdoor activities, it’s a good idea to spend time interacting with the soil. Planting and tending flower beds, fruit trees and a vegetable garden are great ways to get your hands in the dirt on a regular basis. Growing your own fruits and vegetables will naturally introduce potentially healthy microbes to your diet through the trace amounts of soil left on harvested produce. If you are unable to garden, purchasing organic fruits and vegetables from your local farmers market will also promote healthy soil exposure.
Whatever direction you go, banish any thoughts you may have about dirt being bad for you and your kids. As implied through the discovery of malacidins, children (and adults) most certainly will benefit more from playing outdoors and getting dirty than spending long days indoors in front of computers and other electronic devices.








